Steroid implants can be very effective for uveitis, but they are not all the same. Ozurdex is a shorter-acting dexamethasone implant often used when doctors want a stronger but temporary effect. Yutiq is a tiny fluocinolone implant designed to release medicine for much longer and reduce repeat flares over time. Both can help control inflammation, but both can also raise eye pressure and speed up cataract formation. The right choice depends on where the inflammation is, how often it comes back, whether one or both eyes are involved, and your personal glaucoma risk.
Steroid Implants for Uveitis: Ozurdex vs Yutiq Questions Real Patients Ask Before Saying Yes
When patients hear the words “steroid implant,” the first reaction is often mixed. There is relief that there may be a treatment strong enough to calm the inflammation. There is also worry. How long will it stay in my eye? Will it really stop the flares? Am I signing up for glaucoma or cataract surgery later?
These are sensible questions. Uveitis is not a simple disease. It is inflammation inside the eye, and when it keeps coming back or does not settle properly, it can damage delicate tissues that are essential for vision. That is why treatment decisions are often about more than comfort. They are about protecting sight. (FDA Access Data)
In clinic, and certainly in patient forums, the same comparison comes up again and again: Ozurdex or Yutiq? Both are steroid implants placed inside the eye. Both aim to control inflammation locally, meaning the medicine is delivered right where it is needed. But they behave very differently. Ozurdex is a dexamethasone implant that gives a shorter burst of treatment. Yutiq is a fluocinolone implant designed to release medication slowly over a much longer period. (FDA Access Data)
That difference in timing shapes nearly every real-world question patients ask. Let us go through those questions in plain language.
First, what is the basic difference between Ozurdex and Yutiq?
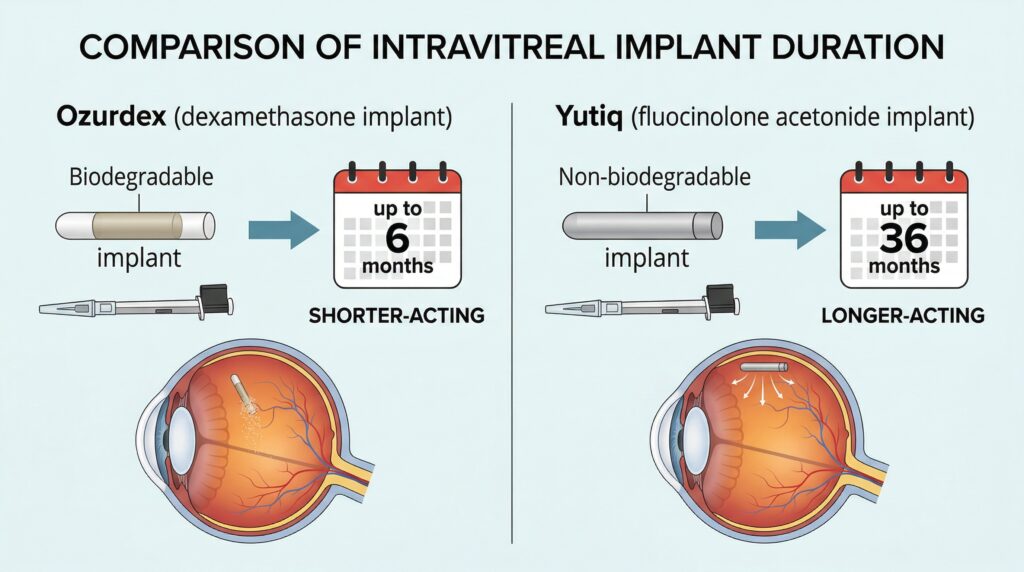
Ozurdex is a biodegradable dexamethasone implant. In simple terms, it is meant to give a stronger short-term anti-inflammatory effect and then gradually break down. In its prescribing information, Ozurdex is approved for non-infectious uveitis affecting the posterior segment of the eye. The label notes that the treatment effect appears within the first two months in many patients, and the duration of effect after onset is roughly one to three months. (FDA Access Data)
Yutiq is different. It is a non-bioerodible fluocinolone acetonide implant, meaning it does not dissolve the same way and is designed to release steroid slowly over a long period. The FDA label states that it lasts 36 months and is indicated for chronic non-infectious uveitis affecting the posterior segment of the eye. That makes it a very different conversation from Ozurdex. It is not mainly about a short rescue effect. It is about long-term suppression of recurrent inflammation. (FDA Access Data)
So the simplest way to think about them is this: Ozurdex is often the “shorter-acting local steroid” option, while Yutiq is the “longer-acting local steroid” option. That is an oversimplification, but for patients deciding whether to say yes, it is a useful starting point. (FDA Access Data)
“Which one lasts longer?”
This is usually the first question, and it matters because most patients are tired of the cycle of flare, treatment, improvement, and flare again.
Yutiq clearly lasts longer. Its label describes a drug delivery system designed to last 36 months. Ozurdex, by contrast, has a much shorter treatment window. In the label, the duration of effect is described as approximately one to three months after onset of effect, although in real practice your own ophthalmologist may discuss a somewhat wider practical window depending on how your eye behaves. The key point is that Ozurdex is not a three-year implant, and Yutiq is specifically designed to be long acting. (FDA Access Data)
This is why some doctors use Ozurdex when they want to see how your eye responds to local steroid treatment before committing to a longer-acting implant. It can function as a way to learn two important things: does your inflammation improve, and does your eye pressure spike when exposed to steroid inside the eye? That second point is crucial. A good anti-inflammatory response is only half the story if the pressure response is dangerous. This stepwise logic is a common clinical way of thinking, although the decision is individualized. (FDA Access Data)
“Which one is better for preventing flares?”
If the problem is recurrent posterior uveitis and the question is long-term flare prevention, Yutiq has the advantage of being built for that purpose. The FDA summary review reported that in two studies, the recurrence rate of chronic non-infectious uveitis within 6 months was significantly lower with Yutiq than with sham injection: 18% versus 79% in one study, and 22% versus 54% in another. That is the kind of data that explains why patients with repeated flares are interested in it. (FDA Access Data)
Ozurdex also has evidence of efficacy in posterior uveitis, but the pattern is different. In the Ozurdex study for posterior segment uveitis, after a single injection, 47% of treated patients reached a vitreous haze score of 0 at week 8 compared with 12% in the sham group, and 43% achieved a three-line visual acuity improvement versus 7% with sham. Those are impressive short-term results, especially when inflammation needs to be brought under control quickly. (FDA Access Data)
So when patients ask, “Which one is better for flare control?” the honest answer is: it depends on what you mean by control. If you mean a shorter-term calming of active inflammation, Ozurdex can be very effective. If you mean long-term reduction of recurrent flares in chronic posterior uveitis, Yutiq is specifically designed for that job. These are not identical use cases. (FDA Access Data)
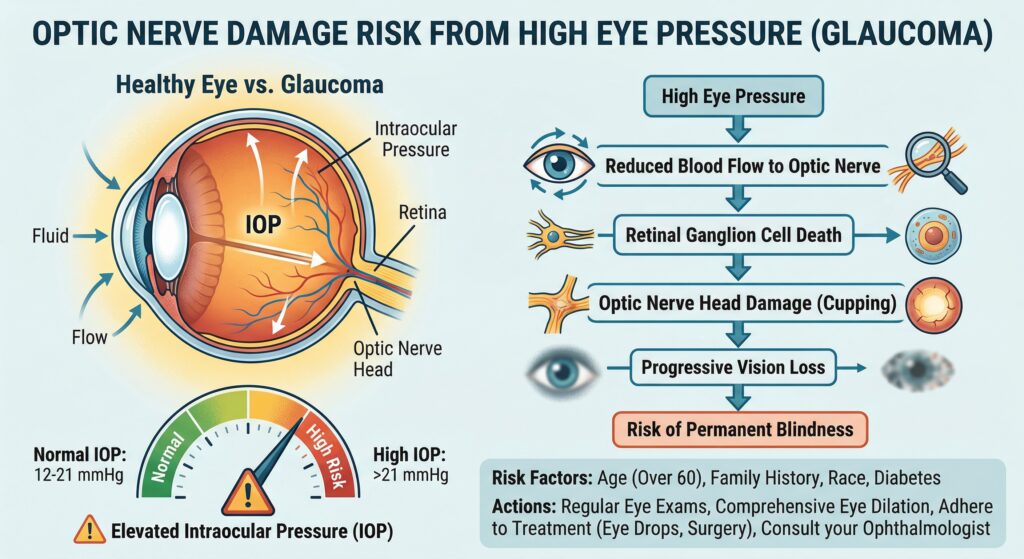
“Am I more likely to get glaucoma from one of these?”
This is one of the most important questions, because steroid response is real. Some eyes tolerate steroids beautifully. Others respond with a meaningful rise in intraocular pressure, and that pressure can threaten the optic nerve if not managed properly.
For Ozurdex, the label reports that in controlled studies, 28% of patients had an intraocular pressure rise of at least 10 mmHg from baseline at any visit, 15% reached at least 30 mmHg, 42% needed pressure-lowering medication, and 1.2% required surgical intervention for elevated pressure. The label also notes that pressure increase peaked at about week 8. (FDA Access Data)
For Yutiq, the label reports that 22% had an intraocular pressure rise of at least 10 mmHg from baseline, 12% reached more than 30 mmHg, 43% used pressure-lowering medication, and 2% needed surgical intervention for elevated pressure. (FDA Access Data)
A few cautions matter here. First, these numbers come from different studies, so they are not a perfect head-to-head comparison. Second, the real issue is not only which percentage looks larger on paper. The more practical issue is your eye’s personal steroid response. If you already have glaucoma, ocular hypertension, suspicious optic nerves, or a strong history of steroid pressure response, your ophthalmologist will weigh that very seriously before recommending a long-acting steroid implant.
In other words, yes, glaucoma risk is part of the conversation for both implants. No responsible specialist should brush that aside. Monitoring after implantation is not optional. It is part of the treatment. (FDA Access Data)
“What about cataracts?”
This is the second major tradeoff. Steroids inside the eye can accelerate cataract formation, especially in people who still have their natural lens.
For Ozurdex, among phakic patients in controlled studies, cataract development was reported in 68% versus 21% in the sham group, and 61% of Ozurdex patients versus 8% of sham patients underwent cataract surgery. (FDA Access Data)
For Yutiq, among phakic study eyes at baseline, cataract was reported in 56% versus 23% in the sham group. (FDA Access Data)
Again, this is not a clean head-to-head comparison because the trials are different. But the broad message is easy to understand: both implants can increase the chance that a cataract appears or worsens over time. That does not mean the implant is a bad choice. In some patients, preserving the retina and controlling inflammation are the priority, and cataract surgery later is an acceptable tradeoff. But it is a tradeoff, and patients deserve to hear that clearly.
“If Yutiq lasts so long, why not just choose it every time?”
Because longer is not automatically better.
A long-acting implant is attractive when uveitis is chronic, recurrent, and mainly in the posterior segment, especially if the eye has already shown it benefits from local steroid therapy. But a longer-acting implant also means a longer exposure to steroid. If you are a strong steroid responder with pressure spikes, a short-acting option may be safer as a trial, or your doctor may move away from local steroid strategies altogether. (FDA Access Data)
There is also a diagnostic issue. Sometimes doctors are still learning the exact behavior of your disease. Is this a one-off severe flare, or a repeatedly relapsing condition? Is only one eye involved? Is there an underlying systemic autoimmune disease? Those answers matter. A long-acting local implant makes the most sense when the disease pattern supports it. (NICE)
“How do implants compare with biologics or oral immunosuppression?”
This is where forum discussions often become confusing, because people compare treatments that are solving slightly different problems.
Steroid implants are local therapy. They treat the eye directly and can reduce exposure to oral steroids and some body-wide side effects. That is one of their big advantages. But they do not treat inflammation elsewhere in the body. If your uveitis is linked to a systemic inflammatory disease, or both eyes are significantly involved, systemic therapy may make more sense because it addresses the whole immune problem rather than one eye at a time. NICE guidance, for example, recommends adalimumab in adults with non-infectious posterior uveitis when there is active disease, inadequate response or intolerance to immunosuppressants, and systemic disease or bilateral involvement with high risk of vision loss. (NICE)
The Multicenter Uveitis Steroid Treatment Trial is also important here, but patients should understand what it did and did not study. MUST compared systemic anti-inflammatory therapy with a fluocinolone implant, but it involved an older implant platform rather than Yutiq specifically. At 24 months, visual acuity improved in both groups, and neither strategy proved clearly superior in visual acuity to the degree detectable by the study’s power. The authors concluded that the advantages and disadvantages of each approach should guide treatment selection for the individual patient. (pubmed.ncbi.nlm.nih.gov)
That is an important message. There is no universal winner. If one eye is the main problem and you want to avoid systemic treatment, an implant may be very reasonable. If both eyes are active, or the disease is part of a larger autoimmune condition, immunomodulatory therapy or a biologic may be the more logical long-term plan. In practice, many patients also receive combinations over time rather than a single forever-treatment. (PubMed)
“Is an implant a sign that my disease is severe?”
Not necessarily. It usually means your doctor is trying to match the treatment intensity and delivery method to the pattern of your disease.
Some patients need an implant because drops cannot reach the back of the eye well enough. Some need it because oral steroids are causing unacceptable side effects. Some need it because the uveitis keeps recurring and a local sustained treatment may protect vision more reliably. And some patients do better with an implant as part of a broader plan that also includes systemic therapy. The word “implant” sounds dramatic, but in uveitis care it is simply one tool among several. (FDA Access Data)
“What follow-up should I expect after saying yes?”
You should expect monitoring, especially for eye pressure, inflammation control, and lens changes. Both FDA labels warn about pressure elevation, infection-related complications of intravitreal injection, and other steroid-related effects, and both recommend patient monitoring after the injection procedure. (FDA Access Data)
In plain language, that means you are not done after the procedure. Your doctor needs to check that the implant is helping, that your pressure is safe, and that no injection-related complication has occurred. Patients who live far away or know they struggle with follow-up should mention that before treatment, because a good implant choice still requires good follow-up care. (FDA Access Data)
“What are the red flags after the procedure?”
Call your eye doctor promptly if you develop worsening pain, rapidly worsening vision, marked redness, increasing light sensitivity, or new flashes and floaters that seem dramatic. Intravitreal injections carry a small but serious risk of infection and other complications, so new severe symptoms after the procedure should not be brushed off as “probably normal.” Both Ozurdex and Yutiq labeling warn about endophthalmitis, inflammation, pressure changes, and retinal complications after intravitreal injection. (FDA Access Data)
So which implant is “better”?
The better implant is the one that fits the disease in front of us, not the one with the louder reputation online.
Ozurdex often makes sense when the goal is fast, local control and a shorter acting steroid effect. It is also useful when your specialist wants to test how your eye behaves with an intravitreal steroid before considering a longer-acting strategy. Yutiq often makes sense when posterior uveitis is chronic or recurrent and the main need is long-term suppression of flares. Both demand respect for the risks of pressure rise and cataract progression. (FDA Access Data)
The real decision is not “Which implant is the best on the internet?” It is “Which treatment plan gives me the best balance of flare control, vision protection, follow-up practicality, and safety in my specific eyes?” That is the right question to take into your appointment. (PubMed)
Questions worth asking your ophthalmologist before you say yes
A good consent discussion should include the following in plain language: Why this implant and not the other one? How long do you expect it to work in my case? What is my personal glaucoma risk? Am I likely to need cataract surgery sooner because of this? If this is only treating one eye, what is the plan if the other eye becomes active? And if the disease is chronic, are we using this instead of systemic treatment, or alongside it? Those questions usually lead to a much better decision than asking for a single “best” option. (FDA Access Data)

When to See a Doctor Urgently
If you have uveitis and notice a sudden drop in vision, severe eye pain, increasing redness, light sensitivity, or a shower of new floaters, do not wait. Those symptoms can reflect an active flare or, after an injection, a complication that needs urgent attention. Uveitis can permanently affect vision if inflammation is not controlled. (FDA Access Data)
Call to Action
If you are deciding between Ozurdex, Yutiq, or systemic treatment for uveitis, book an appointment with a uveitis specialist or ophthalmologist who regularly manages inflammatory eye disease. Bring your medication history, pressure history, and a written list of questions. A careful, individualized plan is far more valuable than a generic online comparison. (PubMed)
References
U.S. FDA Prescribing Information: OZURDEX (dexamethasone intravitreal implant) (FDA Access Data)
U.S. FDA Prescribing Information: YUTIQ (fluocinolone acetonide intravitreal implant) (FDA Access Data)
U.S. FDA Summary Review: Yutiq approval review for chronic non-infectious posterior uveitis (FDA Access Data)
PubMed: Randomized comparison of systemic anti-inflammatory therapy versus fluocinolone acetonide implant for intermediate, posterior, and panuveitis: the MUST trial (PubMed)
NICE Guidance: Adalimumab and dexamethasone for treating non-infectious uveitis (NICE)