Humira (adalimumab) is a biologic medicine used when noninfectious uveitis keeps coming back, involves deeper parts of the eye such as the retina or retinal blood vessels, or cannot be controlled safely with steroid drops alone. The goal is not simply to “use a stronger drug,” but to protect vision while reducing long-term steroid exposure. Patients usually want to know four things first: why now, how well it works, how it is taken, and what safety checks are needed before starting.
Humira for Uveitis: When Patients Move Beyond Steroid Drops and What They Want to Know First
For many patients, uveitis treatment begins in a familiar way: steroid eye drops, frequent follow-up visits, and the hope that the inflammation will settle and stay quiet. Sometimes that works well. But in real clinical practice, many patients reach a point where drops are no longer enough. The eye may flare again as soon as steroids are reduced. The inflammation may sit deeper in the eye, where drops do not reach well. Or the disease may threaten the retina, the retinal blood vessels, or the macula, making long-term vision protection the real priority. That is the point where conversations about Humira, also called adalimumab, often begin. (American Academy of Ophthalmology)
This step can feel emotionally big. Patients often hear the words “biologic” or “immunosuppressive treatment” and immediately worry that the disease must be severe, or that the medicine itself will be overwhelming. In truth, the decision is usually less dramatic and more practical than it sounds. The reason to move beyond steroid drops is often simple: the doctor is trying to control inflammation well enough to preserve vision while avoiding the damage that repeated or prolonged steroid use can cause. (American Academy of Ophthalmology)
Why steroid drops are sometimes not enough
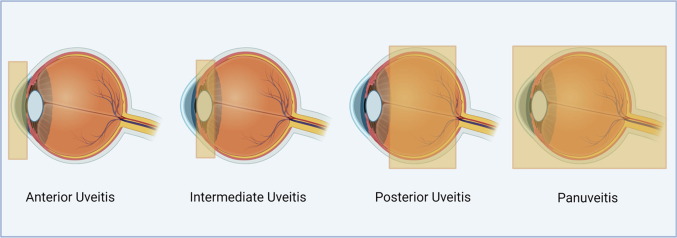
Steroid drops are excellent for many cases of anterior uveitis, where inflammation is mainly in the front of the eye. But they have limits. They do not adequately treat all forms of intermediate uveitis, posterior uveitis, panuveitis, or retinal vasculitis. They also become a poor long-term strategy when inflammation keeps returning, when higher and higher doses are needed to stay quiet, or when steroid side effects begin to appear. (FDA Access Data)
Those side effects matter. Steroids can raise eye pressure, increase the risk of glaucoma, speed up cataract formation, and create a cycle where each flare leads to more treatment and more treatment leads to more risk. In other words, “responding to steroids” is not the same as “being well controlled.” A patient may improve on drops and still be headed toward a pattern that is not safe or sustainable over time. (American Academy of Ophthalmology)
This is especially important in patients with recurrent disease. If inflammation repeatedly returns when drops are tapered, that is a sign the underlying immune process is still active. In patients with retinal vasculitis, cystoid macular edema, or inflammation involving the back of the eye, the issue is even more urgent because the tissues responsible for detailed vision can be damaged quietly, sometimes before the patient notices major symptoms. (American Academy of Ophthalmology)
So where does Humira fit in?
Humira is the brand name for adalimumab, a medicine that blocks tumor necrosis factor alpha, often shortened to TNF-alpha. TNF-alpha is one of the immune system signals that can drive inflammation. By reducing that signal, adalimumab can help calm noninfectious uveitis and lower the risk of flare while steroid treatment is reduced. (American College of Rheumatology)
The US FDA indication for Humira in uveitis is for noninfectious intermediate, posterior, and panuveitis. Current labeling also includes pediatric patients 2 years and older, although adult treatment remains the situation most patients ask about in everyday clinic discussions. (FDA Access Data)
That wording matters. Humira is not used for every kind of eye inflammation. It is used for noninfectious uveitis. Before starting any immunosuppressive treatment, doctors need to be confident that the inflammation is not being caused by an infection such as tuberculosis, herpes viruses, syphilis, or another infectious trigger. Suppressing the immune system in infectious uveitis can make the problem worse, not better. (American Academy of Ophthalmology)
The real reasons doctors bring up Humira
When patients ask, “Why are we talking about injections now?” the answer is usually one or more of these reasons.
The first is recurrent uveitis. If the eye gets better on steroids but relapses each time treatment is tapered, the disease is telling us that short-term rescue therapy is no longer enough. The second is steroid-sparing. This means the doctor wants to control inflammation while reducing the need for long-term steroid drops, oral steroids, or steroid injections. The third is anatomy. If inflammation is in the middle or back of the eye, drops alone may not be the right tool. The fourth is disease severity, including retinal vasculitis or macular involvement, where the cost of repeated flares can be permanent visual loss. (American Academy of Ophthalmology)
In many uveitis practices, systemic treatment is not seen as a “last resort.” It is part of a long-term vision-preserving strategy when the disease pattern shows that local steroid treatment alone is not enough. AAO educational material notes that steroid-sparing drugs are an integral part of long-term management for noninfectious uveitis, and expert guidance emphasizes avoiding long-term corticosteroid exposure when possible. (American Academy of Ophthalmology)
What patients usually want to know first
In clinic, most patients do not start by asking about cytokines or trial design. They ask practical questions.
They want to know, “Why now?” The answer is usually that the pattern of disease has changed from a short flare problem to a long-term control problem. They want to know, “Is this because my case is severe?” Sometimes yes, but often the better word is persistent or recurrent. They want to know, “Will this replace steroids?” Usually the goal is to reduce steroids, not stop them on day one. Many patients begin Humira while steroids are tapered in a structured way. (New England Journal of Medicine)
They also ask, “Does it work?” For many patients with noninfectious intermediate, posterior, and panuveitis, the answer is yes, it can meaningfully reduce the risk of treatment failure and flare. In the VISUAL I trial, adalimumab lowered the risk of uveitic flare or visual impairment in patients with active noninfectious uveitis who were tapered off steroids. In VISUAL II, it significantly lowered the risk of flare or loss of visual acuity in patients whose disease was inactive but steroid-dependent at enrollment. (New England Journal of Medicine)
That is an important point for patients with recurrent disease. Humira is not only for eyes that are inflamed on the day treatment starts. It can also be part of a prevention strategy in patients whose disease becomes quiet on steroids but repeatedly returns when steroids are withdrawn. (American Academy of Ophthalmology)
How Humira is taken
Humira is given as a subcutaneous injection, meaning an injection under the skin, not into the eye. For adult uveitis, the FDA-approved dosing for adalimumab products is an initial 80 mg dose followed by 40 mg every other week starting one week later. Several FDA-approved adalimumab biosimilars use the same adult uveitis dosing in their labeling. (FDA Access Data)
This is one reason many patients find the medicine less intimidating after the first discussion. They often imagine hospital infusions or frequent procedures, but adalimumab is usually a home injection after training. Some patients use a pen device, and others use a prefilled syringe depending on the product and local availability. (FDA Access Data)
The next question is usually, “How soon does it work?” There is no single timeline for every patient. Some begin improving within weeks, but the full treatment plan usually involves overlap with steroid tapering and several follow-up visits. Uveitis specialists judge response not only by symptoms, but also by exam findings, retinal imaging, angiography when needed, and the ability to reduce steroid exposure without a flare. (New England Journal of Medicine)
What safety checks are needed before starting
This is often the most important part of the whole conversation.
Humira suppresses part of the immune system, so infection screening matters before treatment begins. The FDA label warns about serious infections, including tuberculosis, bacterial sepsis, invasive fungal infections, and other opportunistic infections. Patients at risk should be evaluated for prior hepatitis B infection before starting TNF blocker therapy. (FDA Access Data)
In practical terms, many patients will have blood work before treatment and screening for tuberculosis. If there is any concern for latent TB, that needs attention because TNF inhibitors can allow latent TB to reactivate. CDC guidance explains that treating latent TB is highly effective at preventing progression to active TB disease. (CDC)
Doctors also review recent infections, fever, chronic cough, unexplained weight loss, liver history, pregnancy plans, past cancers, neurologic history, and vaccination status. This does not mean most patients cannot take the medicine. It means the decision should be made carefully and safely, with the right baseline checks. (FDA Access Data)

The side effects patients worry about most
Most patients are not as worried about the injection as they are about what the medicine might do to the rest of the body. That concern is understandable.
The common day-to-day side effects include injection-site reactions such as redness, itching, swelling, or discomfort. These are usually mild. More important are the less common but more serious risks, especially infections. Patients are generally advised to contact their doctor promptly for fever, persistent cough, shortness of breath, unusual fatigue, painful skin lesions, or any signs that could suggest a significant infection. (moorfields.nhs.uk)
Another question is vaccines. Patient guidance from rheumatology and the NHS notes that some vaccines are safe while taking anti-TNF therapy, but live vaccines require special discussion and may need to be avoided during treatment. This is one of those details that should be clarified before the first dose rather than after. (American College of Rheumatology)
Patients also ask whether Humira causes cancer. This is a sensitive topic because boxed warnings mention malignancy as well as serious infection. The correct way to discuss this is carefully, not casually. The warning exists, but the individual risk-benefit decision depends on the patient’s age, medical history, other immune diseases, prior treatments, and how much vision is at risk from uncontrolled uveitis. This is exactly why these decisions are made with proper counseling rather than by reading isolated side effects online. (FDA Access Data)
Will I be on Humira forever?
Usually, that is not the first promise a good uveitis specialist makes. The first goal is control. The second is durable control with less steroid exposure. After that, the team looks at how long the eye remains quiet and whether treatment can eventually be reduced. AAO educational material notes that systemic immunosuppression is often continued for around two years after control is achieved, if tolerated medically, although the exact plan varies by disease type and patient response. (American Academy of Ophthalmology)
This is a helpful expectation-setting point. Patients often fear two extremes: either “I will be on this forever” or “I should know in a month whether I’m done.” Neither is usually true. Uveitis treatment is often a medium- to long-term strategy. The timeline is guided by disease behavior, not by impatience or hope alone. (American Academy of Ophthalmology)
What about retinal vasculitis?
Retinal vasculitis is one of the situations that often pushes the conversation beyond drops. When blood vessels in the retina are inflamed, there can be leakage, ischemia, macular swelling, bleeding, and long-term retinal injury. In this setting, the treatment goal is not just symptom relief. It is prevention of cumulative damage. AAO educational material identifies retinal vasculitis among the disease patterns that warrant systemic therapy. (American Academy of Ophthalmology)
Patients with retinal vasculitis often ask whether Humira is being prescribed because the situation is dangerous. The more balanced answer is that retinal vasculitis is important because it can threaten vision, and systemic therapy may offer better control than relying on repeated steroid rescue. The goal is to quiet the blood-vessel inflammation and reduce the chance of repeated flare-related injury. (American Academy of Ophthalmology)
Is Humira always the first steroid-sparing option?
Not always. In many practices, doctors may consider antimetabolite drugs such as methotrexate or mycophenolate as initial steroid-sparing therapy depending on the type of uveitis, associated systemic disease, patient age, pregnancy plans, cost, and local experience. AAO guidance on immunomodulatory therapy and more recent clinical discussions make clear that there is not a single pathway for every patient. (American Academy of Ophthalmology)
However, Humira is an especially important option because it has randomized trial evidence in noninfectious uveitis and an FDA indication for the condition. That gives many patients confidence that the recommendation is not improvised or off-topic. It is a recognized, evidence-based treatment for the right kind of uveitis. (New England Journal of Medicine)
Questions worth asking before you start
If I were speaking directly to a patient in clinic, I would encourage these questions.
Ask what type of uveitis you have and whether it is definitely noninfectious. Ask what the main treatment goal is in your case: fewer flares, less steroid exposure, control of retinal vasculitis, protection of the macula, or all of these together. Ask how your steroid taper will work after the first Humira dose. Ask what screening tests are needed before starting. Ask what symptoms should make you call immediately. And ask how success will be measured, because “feeling better” and “having quiet disease on imaging” are not always the same thing. (FDA Access Data)
The bottom line for patients
Moving from steroid drops to Humira does not mean you have failed treatment. More often, it means your doctor is trying to treat the disease more intelligently and more safely for the long term. In recurrent uveitis, retinal vasculitis, posterior involvement, or steroid-dependent disease, the question is no longer just “What calms this flare?” It becomes “What protects this eye over the next several years?” (American Academy of Ophthalmology)
That shift in thinking is often the hardest part for patients. Steroid drops feel familiar and local. Humira feels bigger and more systemic. But when the disease pattern shows that drops alone cannot safely hold the line, a steroid-sparing biologic may be the more protective option, not the more frightening one. (American Academy of Ophthalmology)
If your ophthalmologist is discussing adalimumab, the most useful first question is not “Is this too strong?” The better question is, “What problem are we trying to prevent that drops can no longer prevent on their own?” Once patients understand that answer, the rest of the conversation usually becomes much clearer. (American Academy of Ophthalmology)

Call to Action
If you or a family member has uveitis that keeps returning, needs repeated steroid treatment, or has been described as retinal vasculitis, intermediate uveitis, posterior uveitis, or panuveitis, book a consultation with a uveitis specialist or ophthalmologist experienced in immunomodulatory therapy. A timely treatment plan can protect vision and reduce avoidable steroid-related side effects. (American Academy of Ophthalmology)
References
- American Academy of Ophthalmology. Strategies for Managing Uveitis, Part 2: Treatment of Adults
https://www.aao.org/eyenet/article/strategies-for-managing-uveitis-part-two - FDA Prescribing Information for HUMIRA (adalimumab)
https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/125057s424lbl.pdf - Jaffe GJ, et al. Adalimumab in Patients with Active Noninfectious Uveitis. New England Journal of Medicine
https://www.nejm.org/doi/full/10.1056/NEJMoa1509852 - American Academy of Ophthalmology. Uveitis Guidelines: Immunomodulatory Therapy
https://www.aao.org/eyenet/article/uveitis-guidelines-immunomodulatory-therapy - American College of Rheumatology. Tumor Necrosis Factor (TNF) Inhibitors
https://rheumatology.org/patients/tumor-necrosis-factor-tnf-inhibitors